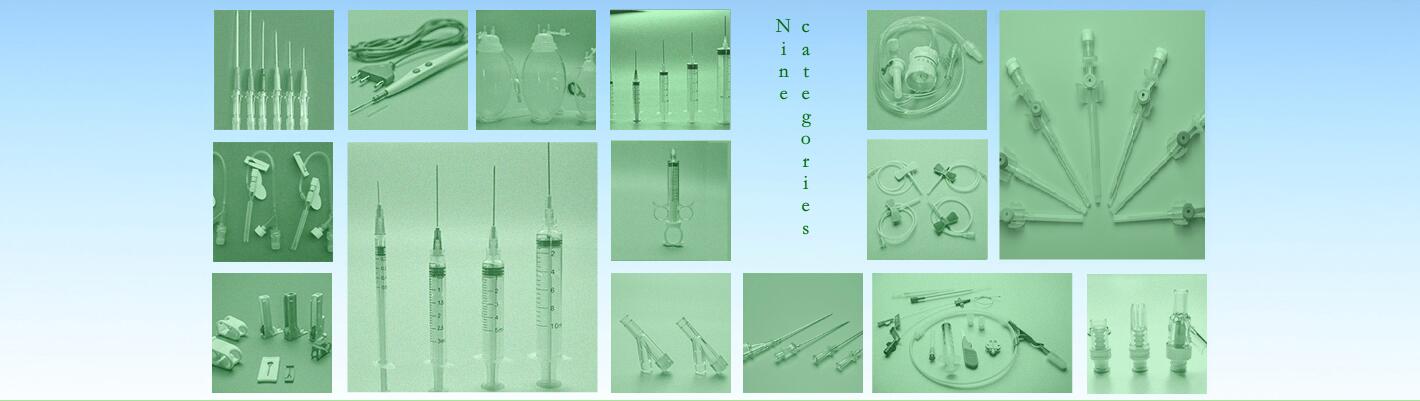
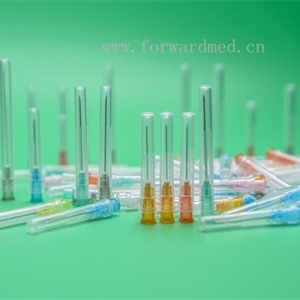
Sterile hypodermic needles for single use
1) intended use
This product is used for human body Subcutaneous, intramuscular or intravenous injection, etc.
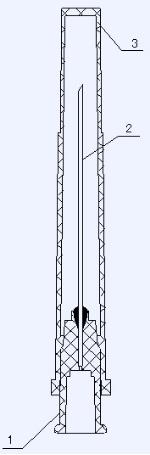
3 | Sheath |
2 | Needle |
1 | Needle base |
NO. | NAME |
4). Classification
ClassⅡa, according to MDD Annex IX, rule 2
5). Product Specification

6) Description of Packaging
8)Contraindications:
No absolute contraindication.
9)Storage:
1. During transportation, the product shall be prevented from great pressure, direct sunlight and rain and snow;
2. The product shall be stored indoors with the humidity no more than 80%, without corrosive gas, cool, dry and well-ventilated.
10)Notice:
1、 Do not use if the package has been previously opened or damaged.
2、 Every access of the store room should keep clean and sterile.
3、 The validity sterilization period is 5 years.